In March 2026, the military procurement website (plap.mil.cn) under the Central Military Commission (CMC)'s Logistics Support Department published disqualification notices against four major pharmaceutical companies in a single day, sending shockwaves through the industry. These four companies – including well-known multinationals and local pharmaceutical companies – were suspended from participating in military procurement across the entire military, with restrictions also extending to other enterprises controlled or managed by their legal representatives and to their authorized representatives. Against the backdrop of both the pharmaceutical industry crackdown and the military’s anti-corruption campaign, legal compliance in interactions between pharmaceutical companies and military hospitals has become particularly critical. This incident serves as a fresh warning: academic collaborations between pharmaceutical companies and military hospitals have entered a new phase where compliance red lines are absolute. This article analyzes the key violation scenarios, penalty standards, substantive impacts on companies, and available remedies. Drawing on typical cases published in recent years, as well as actual cases where we represented companies in dealings with cases related to military hospitals, it examines the specific legal risks and regulatory logic that pharmaceutical companies must consider when participating in military procurement.
I. Under what circumstances will a pharmaceutical company be suspended from military procurement?
In the aforementioned announcement, the four suspended companies were cited for "suspected of violations or dishonest conduct in procurement activities", without disclosing specific reasons. Given that all four companies received identical notices on the same day and all hold leading positions in the anesthesia and psychotropic drug sectors, industry speculation suggests the disqualifications may be linked to a special enforcement action targeting these fields. This area has long been a focus of pharmaceutical anti-corruption efforts: in recent years, public media has reported numerous cases of hospital anesthesia department heads being investigated for receiving bribery. At the medical insurance regulatory level, anesthesia was also included in the national crackdown on medical insurance fraud for 2024 and 2025. Concurrently, the recent investigation of multiple high-ranking military officials reflects an unprecedented anti-corruption resolve within the military system. Military hospitals, as procurement endpoints, are naturally affected. Against this backdrop, whether this action represents the convergence of the special anesthesia sector rectification and military anti-corruption efforts remains to be confirmed by authoritative information.
It is noteworthy that this is not the first time the military has imposed a military-wide procurement ban on pharmaceutical companies. Based on an incomplete statistic of public disqualification records on the military procurement website, as of March 2026, companies in the healthcare sectors have been suspended from military procurement a total of 56 times, involving 53 companies, including several well-known state-owned enterprises. This demonstrates the military’s zero-tolerance approach. Analyzing violation types: bid-rigging and collusive tendering account for over one-third (35.7%); fraudulent bidding/false documentation accounts for 8.9%; 5.4% of cases were referred by military discipline inspection commissions or involved improper benefit transfer to procurement personnel, indicating that the military-local joint anti-corruption mechanism is deeply embedded in procurement supervision processes. Additionally, lending qualifications, contract non-performance, and improper complaints each account for 3.6%. Looking at trends, penalties surged to 35 in 2025 alone – more than triple the total from 2022 to 2024 – and with 18 already recorded in the first quarter of 2026, regulatory scrutiny continues to intensify.
According to the Regulations on the Management of Suppliers for Military Materials, Engineering, and Services (Military Procurement No. 512 [2016]) and related provisions[1], military procurement authorities may impose penalties ranging from written warnings to lifetime bans from military procurement, depending on the nature and severity of the violation. Details are as follows:
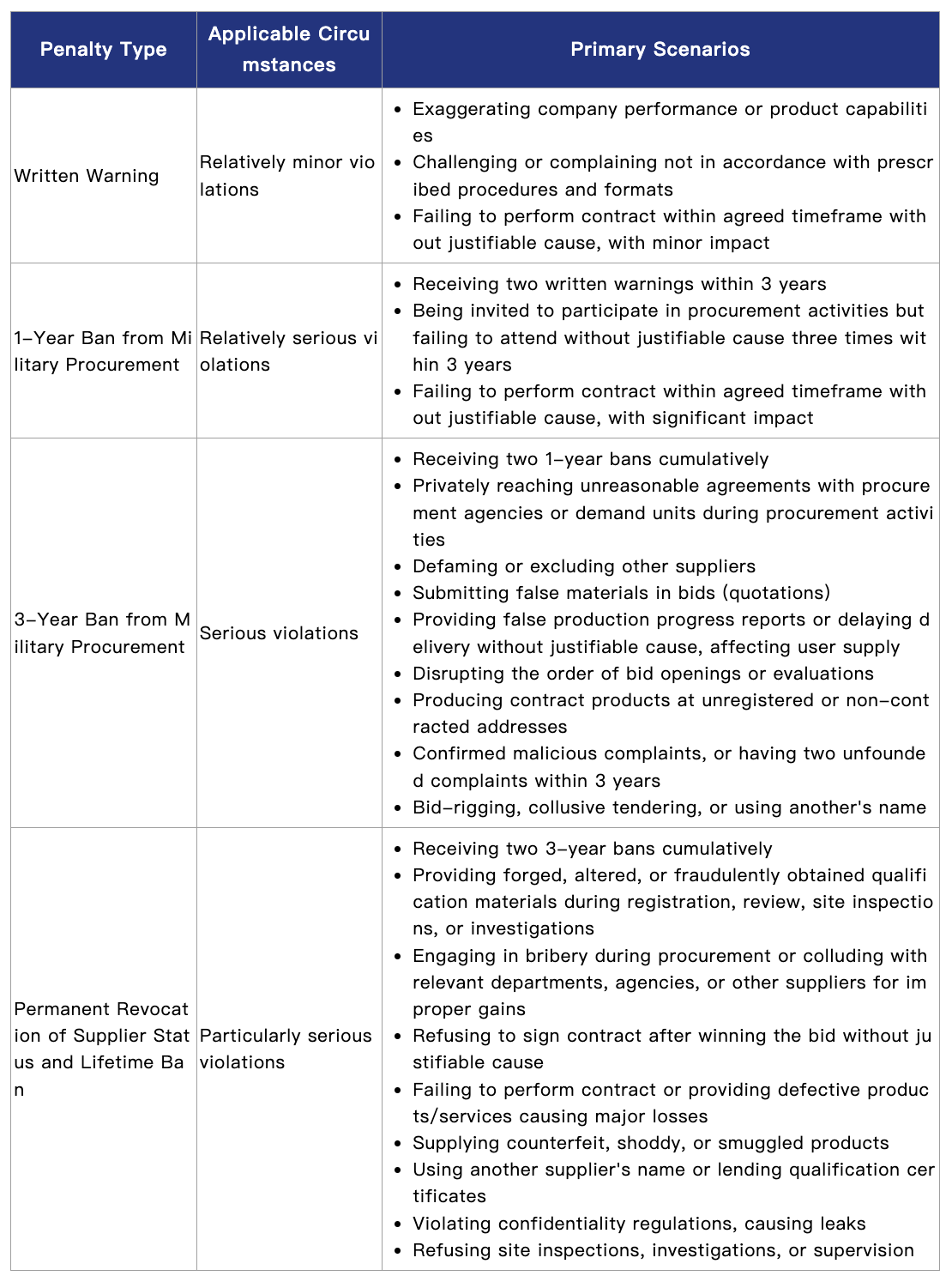
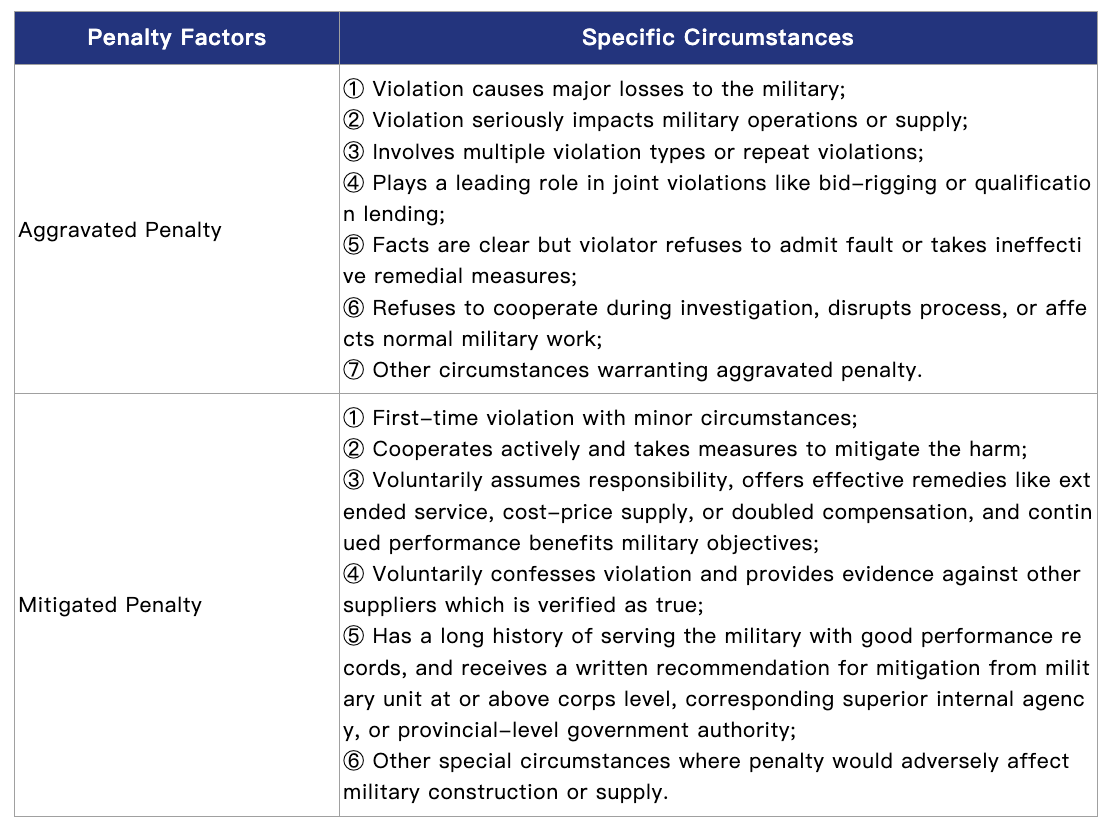
Furthermore, according to the Notice on Clarifying Issues Concerning the Handling of Violations by Suppliers of Military Materials, Engineering, and Services (Military Procurement No. 175 [2022]) and related regulations, military procurement authorities may impose aggravated or mitigated penalties based on specific circumstances.
II. What are the differences and Linkages Between Military Procurement and the Medical Insurance “Credit Evaluation” System?
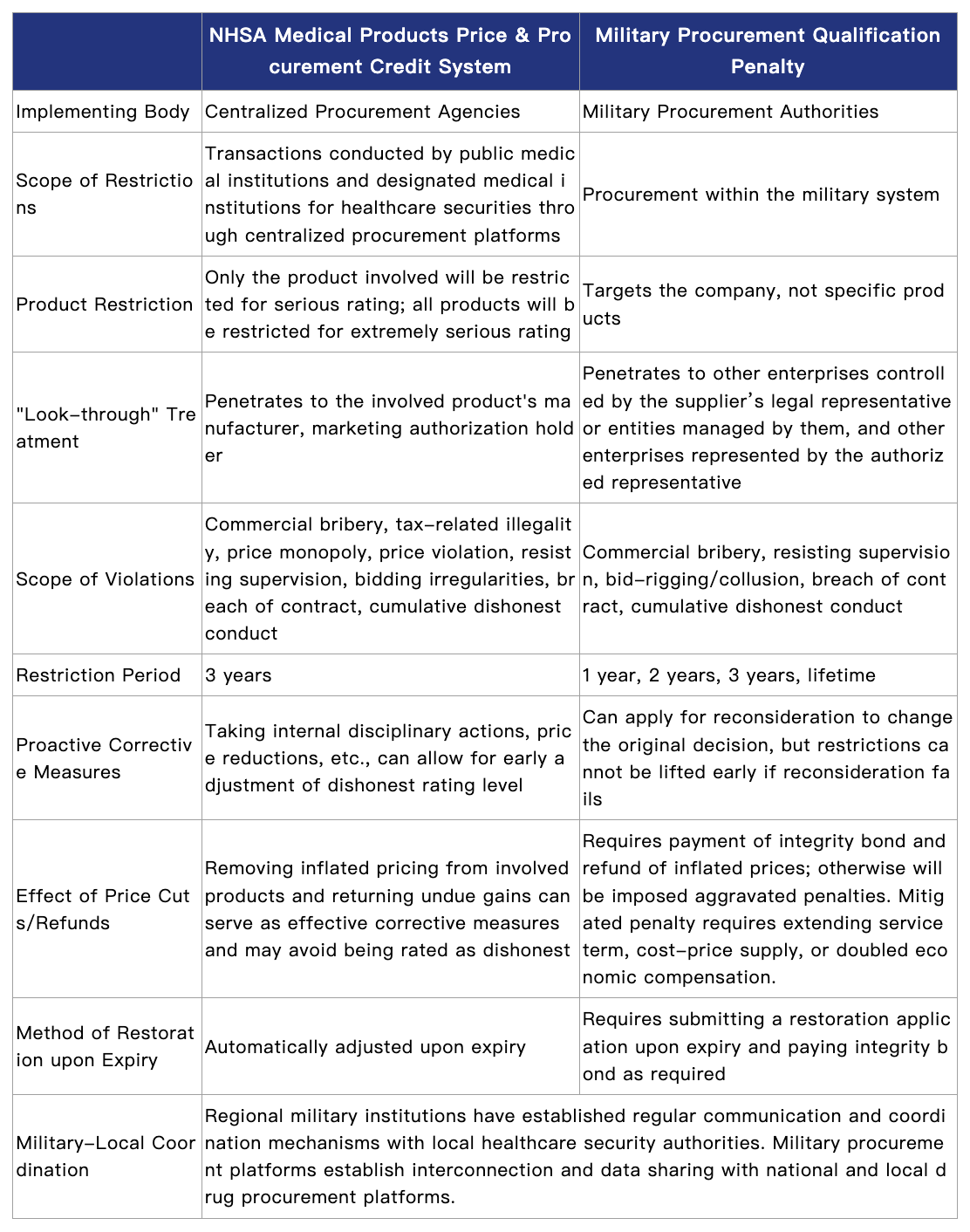
Market access to military hospital procurement is subject to dual management system. Since 2020, the CMC Logistics Support Department and the National Healthcare Security Administration (NHSA) have jointly issued policies for military medical institutions to participate in local drug and medical consumable procurement. Except for combat rescue drugs and consumables procured centrally by the entire military, all other varieties will prioritize local procurement mechanisms. Therefore, local centralized procurement and price bidding credit evaluation systems also apply to military hospitals. Companies must meet both military and local access requirements simultaneously. Key differences are as follows:
Ⅲ. What are the Substantive Impacts on Pharmaceutical Companies Suspended or Banned from Military Procurement?
First, the scope of restriction is extremely broad and does not differentiate by product. As analyzed in Question 2 above, unlike the NHSA’s credit system, military procurement restrictions target the “enterprise as a whole” first and foremost, rather than specific products or categories. Once banned, a company loses eligibility to participate in any material, engineering, or service procurement projects military-wide, an impact far exceeding that of the local procurement system.
Second, joint liability extends to affiliated companies and the legal representative. According to Article 39 of the Regulations on the Management of Suppliers for Military Materials, Engineering, and Services, when a supplier is banned from military procurement, other enterprises controlled or managed by its natural person controlling shareholder or legal representative are also restricted simultaneously; the relevant authorized agent is also barred from representing other suppliers in military procurement. For large pharmaceutical groups, this means one entity's violation can affect multiple subsidiaries within the group. In a recent case, although one involved company was dissolved in 2020, its legal representative was still banned six years later, resulting in restrictions on all other enterprises he controls or manages. In practice, the “legal representative” targeted by penalties usually refers to the individual holding that position at the time of the violation. It is unclear whether the restriction extends to a new employer if the individual, by virtue of having served as the legal representative at the time of the violation, later changes jobs. This uncertainty warrants close attention from pharmaceutical companies.
Third, the military-local joint disciplinary mechanism is now operationally substantive. According to the Notice on Further Strengthening the Supervision and Management of Military Procurement Suppliers and Evaluation Experts (Military Procurement No. 253 [2020]), the military procurement system has established a “military-local joint disciplinary” system. Concurrently, the CMC Logistics Support Department and the NHSA have jointly issued policies clarifying the establishment of policy coordination mechanisms. Regional joint logistics support institutions have established regular communication and coordination mechanisms with local healthcare insurance departments, and military procurement platforms have established interconnection and data sharing with national and local drug procurement platforms[2]. This means that if a company being identified as dishonest in either the local or military system, its eligibility to access the other system may also be adversely affected.
Fourth, companies may face bond recovery and price refunds. According to the Notice on Matters Concerning the Investigation and Handling of Violations by Suppliers of Materials, Engineering, and Services (Military Procurement No. 453 [2024]), if a company violates rules during bidding, military procurement authorities can recover the bid bond within two years after bid opening. If a company is deemed to have “inflated prices” subject to refund and refuses to cooperate, it will face aggravated penalties.
Fifth, restoring procurement qualification requires approval and payment of a bond. Suppliers subject to a ban of less than three years may apply to restore their eligibility to participate in procurement activities after the ban expires. They must pay an integrity bond amounting to 1% to 2% of the project's estimated procurement budget, with a minimum of 100,000 RMB and a maximum of 2,000,000 RMB. Failure to pay will result in non-restoration of eligibility. If payment is not made within six months after the ban period expires, the supplier will be permanently banned. If the supplier receives another ban within three years after restoration, this integrity bond will not be refunded.[3]
Ⅳ. What is the Handling Process of Military Procurement Authorities? What Remedies are Available to Companies?
According to Article 1, Paragraph 3 of the Notice on Clarifying Issues Concerning the Handling of Violations by Suppliers of Military Materials, Engineering, and Services (Military Procurement No. 175 [2022]): “Before deciding to penalize a violating supplier, a notification procedure must be followed. The 'Supplier Notification Letter' should be announced and issued to the supplier in person, informing them of their right to defense within 7 working days; failure to respond within the time limit will be deemed as waiver of the right to defense. If dissatisfied with the penalty decision, an application for reconsideration may be filed within 30 days of the penalty decision; applications submitted after the time limit will not be accepted.” The accepting authority for defense and reconsideration depends on the scope of the penalty. For military-wide penalty decisions, applications should be submitted to the Procurement Service Center of the CMC Logistics Support Department.[4]
It is important to note that currently, there is no clear legal pathway supporting companies in challenging military procurement authorities’ penalty decisions through administrative or civil litigation. Firstly, regarding administrative litigation: although China has established a military administrative litigation system, based on currently public scope of accepted cases, plaintiffs are limited to military personnel or military units[5], excluding suppliers. Furthermore, the military is not considered an administrative organ, so such decisions fall outside the scope of the Administrative Litigation Law. Secondly, according to the Provisions of the Supreme People's Court on Several Issues Concerning the Jurisdiction of Military Courts over Civil Cases, tort or contract disputes where one party is a military unit may be under the jurisdiction of either a military court or a local court. However, qualification restrictions imposed by military procurement authorities on suppliers can hardly be deemed as civil conduct between equal subjects. Currently, there are no publicly reported cases of suppliers suing the military.
Therefore, upon receiving a notification letter or penalty decision, companies must attach great importance to the defense and reconsideration procedures, actively gather evidence, respond promptly, and strive for mitigated penalties, avoiding forfeiting remedies due to procedural missteps.
Ⅴ. Several Issues Requiring Special Attention from Pharmaceutical Companies Participating in Military Hospital Procurement
First, the duality of legal relationships and regulatory authorities. Military medical institutions belong to the military system. Regarding applicable laws, in addition to laws enacted by the National People's Congress and its Standing Committee, they must also comply with military regulations formulated by the CMC or its functional departments, as well as normative documents jointly issued by the military and government. For example, the Measures of the Chinese People's Liberation Army for Implementing the Drug Administration Law of the People's Republic of China was jointly issued by the CMC and the State Council. At the regulatory level, according to the Regulations on the Administration of Medical Institutions, the health authorities of the People's Liberation Army (i.e., the CMC Logistics Support Department and its subordinate units) supervise and manage military medical institutions. Only medical institutions approved by military authorities to provide services to the public can offer medical services to civilians. Notably, in medical insurance fraud cases, because military hospitals need to apply to become designated medical institutions to participate in medical insurance reimbursement, healthcare insurance departments can investigate and penalize military hospitals according to the Social Insurance Law and other regulations.
Second, the division of jurisdiction in criminal cases between military and local authorities. According to the Provisions on Handling Criminal Cases Involving Both Military and Local Jurisdiction and the Trial Regulations on Military Supervision Work, crimes committed by personnel with military status and others performing military tasks are under the jurisdiction of the military supervision commission, military procuratorate, and military court.[6] Cases occurring in non-military management areas of military hospitals[7]are initially investigated by local public security authorities; if the suspect is identified as belonging to the aforementioned military personnel, the case is transferred to military security departments for handling. This means that commercial bribery cases involving military medical personnel may be investigated by the military supervision commission, while medical insurance fraud cases are typically led by local public security authorities, with transfer occurring if military personnel are implicated.
Ⅵ. What are the Compliance Requirements for Pharmaceutical Companies Involved in Military Cooperation Projects?
Military medical institutions are subject to stricter supervision than local hospitals. Following the implementation of the 2016 CMC’s Notice on the Comprehensive Cessation of Paid Services by the Military and Armed Police Forces, military hospitals generally prohibit doctors from directly receiving fees from pharmaceutical companies. Academic visits and conference activities require strict internal approval. Currently, companies often conduct academic exchanges through third-party academic associations to sever direct economic links with individual military personnel.
In daily interactions, pharmaceutical companies must ensure academic promotion strictly adheres to hospital regulations, requires prior appointment and filing, and prohibits any transfer of benefits. Medical device companies need to be cautious that equipment placements avoid bundling clauses linking equipment to consumable purchases, and equipment maintenance services must prevent private dealings with procurement personnel. Additionally, companies should establish special approval processes for military clients, strengthen compliance training and management for KA, sales, and marketing personnel handling military hospitals, and closely monitor military-local joint disciplinary dynamics.
In bidding and market access process, companies must ensure bid documents are absolutely authentic, strictly prohibit providing false qualifications or performance records; maintain independent bidding behavior to strictly prevent bid-rigging and collusion; strictly perform contracts and avoid unauthorized changes to deliverables.
Military medical institutions and military procurement, as vital components of the national healthcare system, directly impact military combat effectiveness and national image. For pharmaceutical companies, participating in military procurement necessitates establishing full-cycle compliance awareness: ensure materials are authentic and maintain independent bidding behavior during the bidding phase; strictly execute contracts during the performance phase; regulate academic exchanges with military medical institutions in daily operations, avoiding any transfer of benefits; simultaneously monitor associated risks related to legal representatives and authorized representatives, as well as the dual risks of military-local joint disciplinary action. Given the strict deadlines for defense and reconsideration in military procurement violation cases, companies must respond promptly upon receiving relevant documents to protect their legitimate rights and interests. Faced with an increasingly stringent regulatory environment, complex compliance requirements, and the continuous improvement of military-local regulatory coordination mechanisms, the compliance threshold for military procurement will be further raised. Only by operating in accordance with the law and performing contracts in good faith can companies secure long-term cooperation opportunities and development space.
Notes:
[1] Several military regulations cited in this article are not publicly available documents. The clauses cited are those publicly referenced by military authorities in relevant announcements.
[2] Ministry of National Defense of the People's Republic of China: "Military Medical and Health Institutions Can Participate in Local Drug and Medical Consumable Procurement," http://www.mod.gov.cn/gfbw/gffw/15950846.html, last accessed March 8, 2026.
[3] Military Procurement Website: "Supplier Registration Instructions," https://www.plap.mil.cn/gpbs-gpbs/#/gpbs/supplierReg/registrationPage, last accessed March 8, 2026.
[4] Military Procurement Website: "Instructions for Applying for Reconsideration of Centralized Military Procurement Projects," https://www.plap.mil.cn/freecms/site/juncai/tzggList/info/2026/2529625.html?noticeType=&channel=95b008df-a4aa-4d77-84fa-bb3f6820f540, last accessed March 8, 2026.
[5] South Sea Fleet: "These Military Administrative Disputes Can Now Be Sued in Military Courts," https://mp.weixin.qq.com/s/eUXK9OHB2uhxDK2sGnkZxw, last accessed March 8, 2026.
[6] Article 4 of the "Notice of the Supreme People's Court, Supreme People's Procuratorate, Ministry of Public Security, Ministry of State Security, Ministry of Justice, and General Political Department of the PLA on Issuing the 'Provisions on Handling Criminal Cases Involving Both Military and Local Jurisdiction'": Investigation, prosecution, and adjudication of military personnel shall be under the jurisdiction of military security departments, military procuratorates, and military courts. Military civilian personnel, non-active duty service staff, civilian staff on the establishment, retirees managed by the military, and reserve personnel and others performing military tasks shall have jurisdiction determined as per military personnel. The "Trial Regulations on Military Supervision Work" stipulate that military supervision commissions supervise the exercise of power by active-duty military personnel, civilian personnel, retirees managed by the military, other personnel, and reserve personnel and others performing military tasks.
[7] Article 5 of the "Notice of the Supreme People's Court, Supreme People's Procuratorate, Ministry of Public Security, Ministry of State Security, Ministry of Justice, and General Political Department of the PLA on Issuing the 'Provisions on Handling Criminal Cases Involving Both Military and Local Jurisdiction'": ...Cases occurring outside military camps shall be investigated and filed by local public security authorities, national security authorities, or people's procuratorates; if the suspect is determined to fall under the jurisdiction stipulated in Article 4, Paragraph 1 of these Provisions, the case shall be transferred to military security departments or military procuratorates for handling. Article 6: ...Cases occurring in offices established by the military in local state organs and units, premises providing services to the public, residential communities under property management, and locations or residences where alert duties are performed locally shall be handled according to Article 5, Paragraph 2 of these Provisions. Article 20: "Military camp" as used in these Provisions refers to areas managed and used by the military, including military restricted zones, military management zones, and temporary military stations, etc.










